Background
Primarily skin aids in shielding our bodies from prospective attacks by foreign organisms and toxic elements. It also acts as an interface with the external environment and is colonized by a diverse assortment of microorganisms – including bacteria, fungi, viruses, and mites. Although disease-causing pathogens are most often and commonly studied organisms, commensal microbes that benefit the host, also have a significant, if perhaps often unappreciated, role in human health.
Microbiology and dermatology are intimately interrelated and it has long been recognized that these microbial populations are indirectly associated with human health and diseases (Beri, 2018). The community of microbes acclimatizes or adapts to its environs, shows the variation in population density determined by various chemical, biological, and physical conditions on the skin such as dry, moist, or sebaceous skin (Fig.1). This in turn could partially be attributed to the density of glands and hair follicles (Bay et al, 2019). Taking into account only the number of bacteria it is estimated that approximately 1 million bacteria with hundreds of distinct species inhabit each square centimeter of skin.


Some of the most commonly found flora on the skin are depicted below –
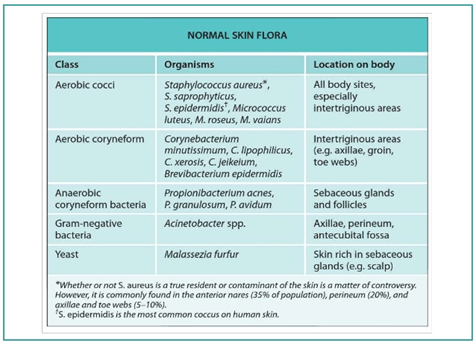
Metagenomic sequencing of the microbiome on various human body sites provides an understanding of the influence of various factors including external milieu, ecosystem, host immunity, genetic susceptibility, and host lifestyle on microbial biodiversity. Microorganisms of the skin extend from the exterior epidermal layer of the skin to the deeper dermis and almost 25% grow in the deeper layer of the dermis and sebaceous glands.
Microbes are further classified as resident and transient, where residents are those which are passed on during birth from mother or picked up from interaction with day to day surroundings (animals, plants, persons, chemicals, climates) and are long-lasting. Any change in the environment leads to the development of novel transient microorganisms groups which are eradicated once back to normalcy.
Thus, four varieties of microbes inhabit the skin – stable residents, transients, temporary residents, and pathogens. Every organism has a unique and exclusive signature of skin microbiota acquired during infancy and stabilized during adulthood. Many of these microorganisms are harmless and in some cases provide vital functions. Symbiotic microorganisms inhabit a wide range of skin niches and shield against invasion by more pathogenic organisms. They also have a role in educating billions of T cells (T lymphocytes of the adaptive immune system) found in the skin by training them to counter pathogens (Grice and Segre, 2011).
The configuration of abundant species is relatively stable over time. However, skin diseases such as acne vulgaris, eczema, psoriasis, or dandruff bring about strong and specific alterations. For instance, the development of acne vulgaris has been associated with dysbiosis (imbalance in natural flora leading to illness) in the skin microbiome, Cutibacterium acnes (Paetzold et al., 2019). Various strains of this bacterium are associated with different degrees of skin-related disorders especially acne. Similarly, several other strains have the ability to produce positive effects; a potential that can be tapped for developing therapeutic strategies for skin disorders by manipulation of these microbes.
Sampling methods
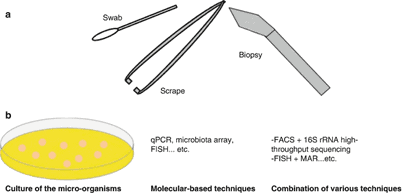
Conventional method – swabbing
Semi quantitative methods – replica plating, sticky tape sampling, roll tubes
Quantitative methods – cylinder, liquid vehicle, scrubbing; Air sampling technique (settle plates, impaction sampler)
Skin Biopsy – Histopathological examination
Molecular approaches to study host – microbe interaction
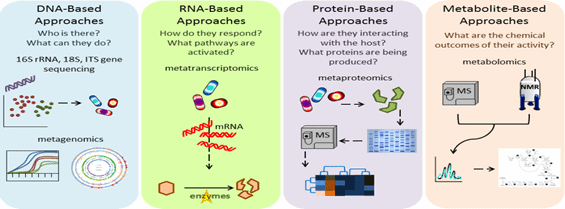
Microbial diversity and composition of non- human vertebrate skin
A lot is recognized and examined by molecular methods to recognize microbiomes on human skin, but skin microorganisms associated with other vertebrates are yet to be explored and studied comprehensively. This, in particular, is important to reveal an association between skin microbes and various vertebrate characteristics such as topographical position, biological sex, animal communications, diet, confinement, maternal transfer, and disease. Reviewing these factors is vital to design future microbiome studies whose outcomes could be extrapolated to develop useful applications, such as probiotics and designing conservation strategies for endangered and threatened animals.

Researchers, through modern technologies and sampling protocols, have identified microflora in several animals. The next step now is to interpret and make use of the outcomes to improve the lives of studied animals.
Developing skin probiotics to protect animals from skin infections threatening the extinction of various species, could be the next target, which will be beneficial from economic point of view as well as for providing healthier food for human consumption. It will also help in planning conservation strategies for endangered organisms. Thus proving favorable to aquaculture industry, animal husbandry and pet industry.
Practical applications of human skin microbiome
Developing a comprehensive therapeutic strategy for the treatment and study of diseases especially those caused by antibiotic-resistant bacteria is the most common application. Likewise, manipulation of skin microbiome has the potential to develop better therapeutic approaches for skin diseases. An improved understanding of the interactions between surface and deeper microbial population and immune cells will help us to design topical cosmetics to be used externally but exerting deeper effects in the dermal tissue and enhancing immune functions of the host through various pathways.
Similarly development of epidermal exfoliating treatments for anti-aging and innovative cosmetics that stimulate a renewal process within the epidermal layer by manipulating local immune signals which govern host regenerative pathways can be done.
With the understanding of dysbiosis and its influence on host immunity and regenerative pathways of the skin, treatment strategies for the most common and stubborn skin conditions such as acne and eczema can be revolutionized.
Another interesting aspect of understanding the relationship between the skin and gut microbes opens avenues to correlate diet and its influence on skin, a fact emphasized in Ayurveda since ages. Vibration science dealing with healing methods reiki and sound vibrations, interrelating the philosophy of interconnectivity of all living creatures through energy can also be validated by exploring the possibilities of manipulating microbiome to influence emotions, through evolution of novel cosmetics triggering local neuroendocrine pathways.
Understanding the skin–brain axis could be vital in assessing any impact of meditations and mindfulness with changes in surface microbiome, in turn improving skin conditions. Various studies correlating breathing and yoga to improvement in overall wellbeing can provide guidance of this axis in a more comprehensive manner and can help associating surface flora changes with antioxidant levels of blood which directly influences health.
More research is required in exploring devices with penetrating active ingredients affecting regenerative and immune pathways in the deeper layers of the skin eliciting potent host response. Finally, study of the microbiome and host immune interaction can help deliver safer and potent cosmetic products. Concept of the ‘cosmetic microbiome’, which can influence the skin–gut–brain relationship can be adapted to design innovative cosmetics and transdermal drugs revolutionizing perception of beauty, health and wellbeing. Thus, cosmetics in the future could have the potential to claim that they can not only make you look good but also ‘feel’ good.
Recommendations
Human skin microbiota especially of epidermal layer is unique and differs as per individual’s lifestyle and genetic predisposition. However the preserved dermal microbial community is compositionally unique and functionally distinct to the specific environment. As it is in direct contact with the immune system of the host, further research for host microbiome interaction should be targeted to this cutaneous compartment. This novel insight into specific microbial adaptation can be used advantageously in the research of chronic disorders and infections of the skin in turn benefitting patients suffering from long-lasting socioeconomic illnesses.
References
Bay, L., Barnes, C.J., Fritz, B.G., Thorsen, J., Restrup, M.E.M., Rasmussen, L., Sørensen, J.K., Hesselvig, A.,B., Odgaard, A., Hansen, A.J., Bjarnsholt, T. (2020). Universal Dermal Microbiome in Human Skin. Amer. Soc. MicroBiol. mBio 11(1): e02945-19.
Beri, K. (2018). Skin microbiome & host immunity: applications in regenerative cosmetics & transdermal drug delivery. Future Sci. OA 04(06):FSO302.
Grice, E.A., Segre, J.A. (2011). The skin microbiome. Nat Rev Microbiol. 9(4): 244–253.
Ibrahim, S., Belheouane, M. (2018). Methods for Microbiota Analysis: Sample Collection and Laboratory Methods. In: Ragab G., Atkinson T., Stoll M. (eds) The Microbiome in Rheumatic Diseases and Infection. Springer, Cham. https://doi.org/10.1007/978-3-319-79026-8_2
Ilhan, Z.E. (2016). Microbiome After Bariatric Surgery and Microbial Insights into Surgical Weight Loss. Ph. D. Disertation, Arizona State University. August 2016.
Kong, H.H., Segre, J.A. (2012). Skin Microbiome: Looking Back to Move Forward. J Investig. Dermatol. 132:933–939.
Paetzold, B., Willis, J.R., de Lima, J.P., Knödlseder, N., , Brüggemann, H., Quist, S.R., Gabaldón, T., Güell, M.(2019). Skin microbiome modulation induced by probiotic solutions. Microbiome 7:95.
Ross, A.A., Hoffmann, A.R., Neufeld, J.D. (2019). The skin microbiome of vertebrates. Microbiome 7:79.















14 Responses
Life has always intrigued me…ur article shows us another way look at the microbial world…
Life is a very interesting Science fact. We will help you to explore more about that. Stay connected, don’t forget to register on our website.
Highly informative. Made me realise that the smallest of things can cause huge effect on our body and we might not even know it. Astounding
Thank you for your comment. Keep supporting us.
Deeply researched and informative article.
Hope you do more research and help people become healthier by understanding microbes better.
We appreciate your comment and we always welcome suggestions. We will definitely cover this topic further in future articles.
Informative and relevant article. Meticulously written. Good research stuff.!
Very interesting and informative research work.. nicely written ..
Thank you so much. Don’t forget to register on our website for further updates.
Thank you so much for your comment. Don’t forget to register on our website.
I’m amazed at the complexity of the human body. Well researched and interesting article.
The human body is a very complex structure, every part of a body plays a vital role. Register to our website for more updates in future.
Very informative.. Keep it up..
Thank you so much for your valuable comment. Don’t forget to register with us, you can download our web application as well.