The planet never fails to amaze us in its ways. And with science, we can understand all the magic that nature throws at us with certain logic. The immortality that we all see in science fiction movies and read in books is a real phenomenon. In recent year’s scientists have found many shreds of evidence of many organisms that show immortality is a real deal.
But there’s a small catch this doesn’t mean that the organisms mentioned in the article will never die. They may die from other causes but will not die of ageing. They can be killed by a predator, disease, or any catastrophic event like volcanic eruptions. This type of immortality is referred to as biological immortality by scientists. By doing specific research on all these organisms scientists all around the world have only one final goal to reach and that is to understand and conquer over immortality. But before these conquests are finished let us decode the science behind the biological immortality of the following organisms one by one.
The ‘immortal’ jellyfish
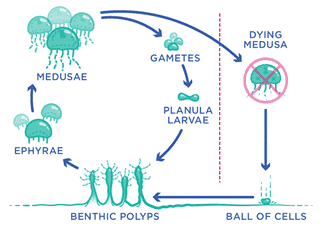
The first organism that was first proved to be immortal is a species of jellyfish called Turritopsis dohrnii. These jellies like small, marine, transparent organisms with stinging tentacles roam around in the depth of oceans are very interesting organisms that have a life cycle that can turn back time. When injured or hurt the species can revert to their earlier phases to recover the damaged phase. Their life cycle compromise of the following steps
- The lifecycle of jellyfish begins with the formation of an egg which is by fertilization of male and female gametes.
- The fertilized egg then develops into small larvae also known as a planula.
- The larvae being motile begin its first swim where it sticks to a rock or any other surface and here the planula develops into a polyp. The polyps generally have a foot that is attached to the surface and are hence non-motile. The jellyfish will remain in this form for some time and will form a colony of polyps.
- After which, one of these polyps will form buds that will eventually get separated from the body. Or sometimes depending on the species it may produce a whole total of a separate segment and break away from the colony.
- For most of the species of jellyfish this is the end of the line but in the case of Turritopsis dohrnii here comes the twist of fate.
- The jellyfish when faces with any environmental stress or natural injuries has always the option of transforming into a polyp again. It’s like a butterfly going back to its caterpillar form or a frog going back to its tadpole form.
This thing that looks like magic is a beauty of nature. Still, we can’t consider it truly immortal because it can still die of a very drastic change of events.
There is also a possibility of many other species of jellyfish lying in the deep sea beds with similar features yet to be discovered.
HYDRA

Hydra is freshwater tiny hydrozoa with stinging tentacles. They look the same as the polyps of jellyfish this is also because they belong to the same phylum (cnidaria). Its one end has to mouth with tentacles surrounding it and the other end has a sticky footer. They are primitive type organisms and spend most of their time in one place in freshwater ponds or rivers. Hydra usually uses their stinging tentacles to catch their prey and put it to their mouth.
The thing that makes them interesting is the fact that they do not go through the phase of senescence at all. Instead of the gradual degradation like other organisms over time, it feels like they go towards healing. A hydra’s stem cell has the unique capacity to self-renew itself, thanks to the pair of Fox0 genes which are found in animals to human and are responsible for the longevity of a cell.
Here in case of hydra, this gene is a bit tweaked. The Fox0 genes are found in much abundance and when these genes are expressed the result comes out to be a special ability which makes them nearly immortal. During an experiment to identify the cause of this mysterious phenomenon the fox0 gene was suppressed which stopped the regeneration and the hydra started to age. Scientists are still unable to know the actual cause of this endless youthfulness.
Immortal lobsters

Lobsters also do not experience senescence but unlike hydra, they do not rely on certain genes but rather they can endlessly repair their DNA.
Normally in every organism during the phase of cell division, the telomeres get short during each division. At last, there comes a stage where the telomeres get the shortest after which more division is not possible and the organism attain the state of senescence. Thankfully lobsters don’t have this problem as they get a regular supply of telomerase enzyme for which they keep regenerating telomeres. They keep producing the enzyme throughout their lives which helps them manage their all-time youthful status.
Every organism found in the earth has telomerase enzyme present in them but most of the enzyme is only being produced only up to their embryonic stage. After passing the embryonic stage the cells in the body lacks the amount of telomerase required to divide eternally.
But their only bliss becomes their curse. As they keep dividing infinitely their body grows but their outer shell remains the same. For this problem, they also keep changing and forming new exoskeletons each time they grow. This process requires a lot of energy and providing this amount of energy for lots of time finally results in starvation and death.
Regenerating worms

The creepy flat crawlers that are also known as planarian worms of phylum Platyhelminthes have a unique ability to regenerate itself. When cut lengthwise or across into two pieces then it can form two new worms. Even if only 1/5th of the worm is cut of still they grow into a new worm. This limitless regeneration ability also applies to ageing and damaged tissues which allows the worms to live indefinitely. This ability makes them technically immortal. According to a study done by the University of Nottingham, the scientist discovered a very astonishing fact that the planarian worms found in marine water can cheat death indefinitely and keep on living for years.
Radiation-resistant bacterium

Last but not least is the most indestructible species on the planet. This type of bacterium is resistant to every chemical and physical thing found on the planet. They can even die and also come back to life thanks to their immediate DNA repair response. According to the Guinness book of the world record, they have been listed as the world’s toughest bacterium. They can survive extremities in heat, cold, salinity, alkalinity, acidity, and even vacuum. They are so tough to kill that they can even resist 1.5 million gamma rays which are 3000 times more than the amount that will kill a human being in an instant.
ATTEMPT TO ENGINEER BIOLOGICAL IMMORTALITY IN HUMANS
The thing always remains to be a controversial topic mostly due to ethnic reasons. Many also believe the thing asked for is not considered to be practical. One of the most important and serious collaborations in this area is being done by the SENS Research Foundation. The organization claims to deliver negligible senescence for human beings using gene therapy and genetic engineering.
Many other efforts were also being made towards this direction –
- In 2015, Elizabeth Parrish, CEO of BioViva, treated herself using gene therapy with the goal of not just halting, but reversing ageing. This effort was widely criticized
- Many organizations used brain preservation to store and preserve the mammalian brain for a longer time using suspended animation technique without the neurons getting damaged.
- In 2016 some doctors of mayo clinic did some experiment to extend the mice life span by 25%. And the same organization is now developing things for human trials.
- A September 2019 article claims to have reversed epigenetic ageing in humans successfully.
- In November 2019 the trials of first human telomere lengthening clinical trial started, this experiment aimed to reverse human ageing least by 20 years. This trial was highly criticized due to the dangers of using a genetically modified virus. the source was also hidden from the general public.
- In May 2020, there was a successful experiment to reduce ageing in mice by an average of 54%, with the transfusion of young blood plasma.
Closure
The study in these all subjects is today also incomplete and there may be many more such organisms lying in the depths yet to be discovered. Scientists and geek all over the world are still searching for pieces of evidence of the animals that can lead us to the secret of pure immortality and till then let us embrace the beauty of these scientific miracles.
REFERENCES:
http://www.bbc.com/earth/story/20150622-can-anything-live-forever
Masoro, E.J. (2006). Austad, S.N. (ed.). Handbook of the Biology of Aging (Sixth ed.). San Diego, CA: Academic Press. ISBN 978-0-12-088387-5.
Michael R. Rose; Casandra L. Rauser; Laurence D. Mueller (Nov–Dec 2005). “Late life: a new frontier for physiology”. Physiological and Biochemical Zoology. 78 (6): 869–878. doi:10.1086/498179. PMID 16228927.
Shay, J. W. & Wright, W. E. (2000). “Hayflick, his limit, and cellular ageing”. Nature Reviews Molecular Cell Biology. 1 (1): 72–76. doi:10.1038/35036093. PMID 11413492.
Skloot, Rebecca (2010). The Immortal Life of Henrietta Lacks. New York: Crown/Random House. ISBN 978-1-4000-5217-2.
Smith, Van (2002-04-17). “The Life, Death, and Life After Death of Henrietta Lacks, Unwitting Heroine of Modern Medical Science”. Baltimore City Paper. Archived from the original on 2004-08-14. Retrieved 2010-03-02.
Bulzomi, Pamela. “The Pro-apoptotic Effect of Quercetin in Cancer Cell Lines Requires ERβ-Dependant Signals.” Cellular Physiology (2012): 1891-898. Web.
Reitzer, Lawrence J.; Wice, Burton M.; Kennel, David (1978), “Evidence That Glutamine, Not Sugar, Is the Major Energy Source for Cultured HeLa Cells”, The Journal of Biological Chemistry, 254 (April 25): 26X9–2676, PMID 429309
University of Cologne (7 March 2018). “On the immortality of stem cells”. ScienceDaily. Retrieved 17 September 2020.
Surani, Azim (1 April 2009). “Germ cells: the route to immortality”. University of Cambridge. Retrieved 17 September 2020.
Michael R. Rose; Casandra L. Rauser; Laurence D. Mueller (1983). “Expression of the Large T Protein of Polyoma Virus Promotes the Establishment in Culture of “Normal” Rodent Fibroblast Cell Lines”. PNAS. 80 (14): 4354–4358. Bibcode:1983PNAS…80.4354R. doi:10.1073/pnas.80.14.4354. PMC 384036. PMID 6308618.
Irfan Maqsood, M.; Matin, M. M.; Bahrami, A. R.; Ghasroldasht, M. M. (2013). “Immortality of cell lines: Challenges and advantages of establishment”. Cell Biology International. 37 (10): 1038–45. doi:10.1002/cbin.10137. PMID 23723166.
Species with Negligible Senescence Archived 2015-04-17 at the Wayback Machine. AnAge: The Animal Ageing and Longevity Database
“Calico Scientists Publish Paper in eLife Demonstrating that the Naked Mole Rat’s Risk of Death Does Not Increase With Age”. Calico. 25 January 2018. Archived from the original on 27 January 2018. Retrieved 27 January 2018.
“Naked mole rats defy the biological law of aging”. Science Magazine – AAAS. 26 January 2018. Archived from the original on 26 January 2018. Retrieved 27 January 2018.
Ruby, Graham; Smith, Megan; Buffenstein, Rochelle (25 January 2018). “Naked mole-rat mortality rates defy Gompertzian laws by not increasing with age”. eLife. 7. doi:10.7554/eLife.31157. PMC 5783610. PMID 29364116.
Current Biology: Volume 23, Issue 19, 7 October 2013, Pages 1844–1852 “Fission Yeast Does Not Age under Favorable Conditions, but Does So after Stress.” Miguel Coelho1, 4, Aygül Dereli1, Anett Haese1, Sebastian Kühn2, Liliana Malinovska1, Morgan E. DeSantis3, James Shorter3, Simon Alberti1, Thilo Gross2, 5, Iva M. Tolić-Nørrelykke1
Martínez, Daniel E. (1998). “Mortality patterns suggest lack of senescence in Hydra” (PDF). Experimental Gerontology. 33 (3): 217–225. CiteSeerX 10.1.1.500.9508. doi:10.1016/S0531-5565(97)00113-7. PMID 9615920. Archived (PDF) from the original on 2016-04-26.
De Vito; et al. (2006). “Evidence of reverse development in Leptomedusae (Cnidaria, Hydrozoa): the case of Laodicea undulata (Forbes and Goodsir 1851)”. Marine Biology. 149 (2): 339–346. doi:10.1007/s00227-005-0182-3. S2CID 84325535.
He; et al. (2015-12-21). “Life Cycle Reversal in Aurelia sp.1 (Cnidaria, Scyphozoa)”. PLOS ONE. 10 (12): e0145314. Bibcode:2015PLoSO..1045314H. doi:10.1371/journal.pone.0145314. PMC 4687044. PMID 26690755.
Cong YS (2002). “Human Telomerase and Its Regulation”. Microbiology and Molecular Biology Reviews. 66 (3): 407–425. doi:10.1128/MMBR.66.3.407-425.2002. PMC 120798. PMID 12208997.
Wolfram Klapper; Karen Kühne; Kumud K. Singh; Klaus Heidorn; Reza Parwaresch & Guido Krupp (1998). “Longevity of lobsters is linked to ubiquitous telomerase expression”. FEBS Letters. 439 (1–2): 143–146. doi:10.1016/S0014-5793(98)01357-X. PMID 9849895.
Jacob Silverman (2007-07-05). “Is there a 400 pound lobster out there?”. howstuffworks. Archived from the original on 2011-07-27.
David Foster Wallace (2005). “Consider the Lobster”. Consider the Lobster and Other Essays. Little, Brown & Company. ISBN 978-0-316-15611-0. Archived from the original on October 12, 2010.
IMAGE REFERENCES
http://www.bbc.com/earth/story/20150622-can-anything-live-forever














One Response