Recently a pig kidney is transplanted into a human without any significant immediate rejection by the recipient’s system. The whole process was done at NYU Langone Health in New York City involving the use of a pig whose genes had been altered so that its tissues no longer contain a molecule responsible for triggering instant rejection.
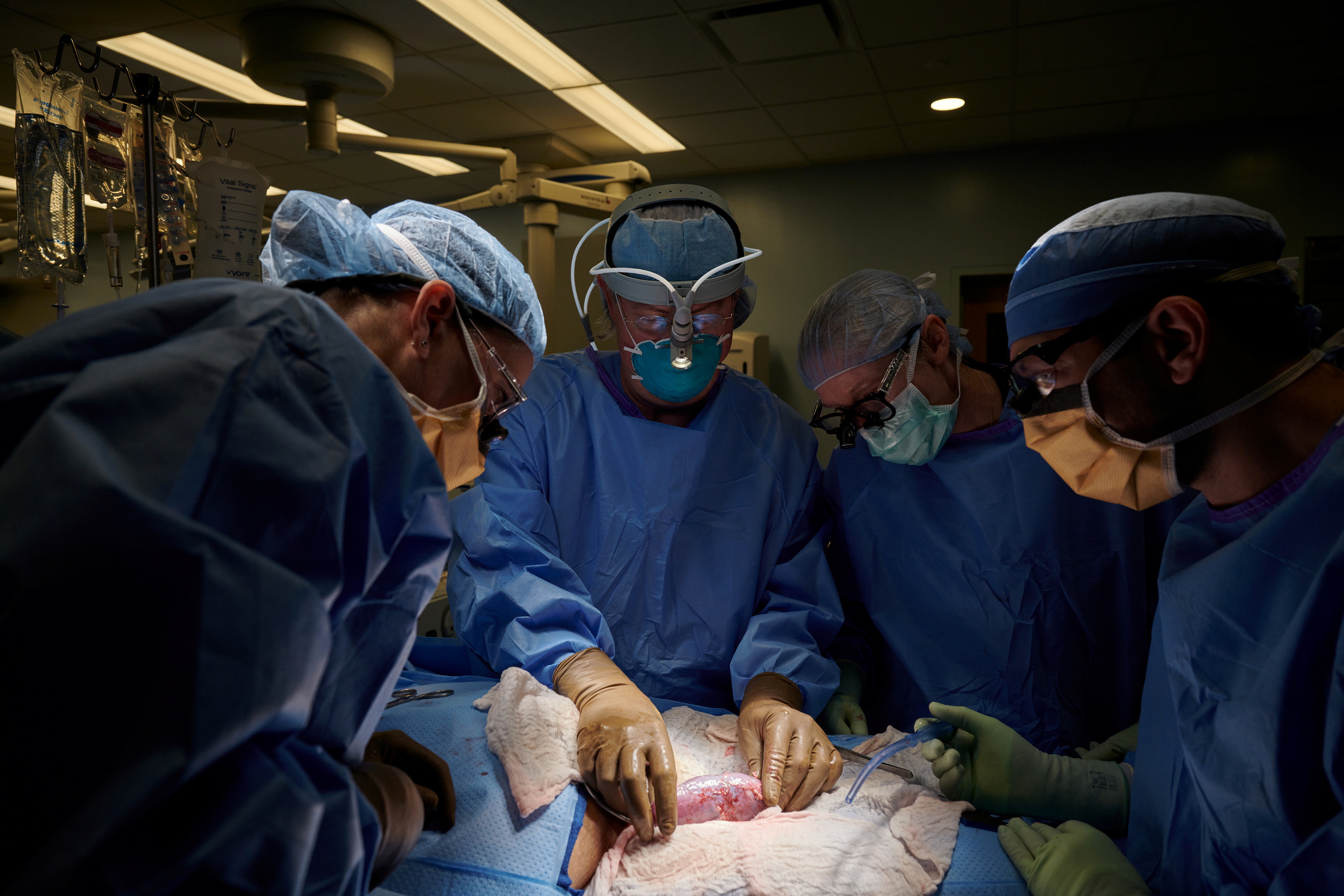
The surgical team examines the pig kidney for any signs of hyperacute rejection, as the organ was implanted outside the body to allow for observation and tissue sampling during the 54-hour study period, at NYU Langone in New York, U.S., in this undated handout photo. Joe Carrotta for NYU Langone Health/Handout via REUTERS .
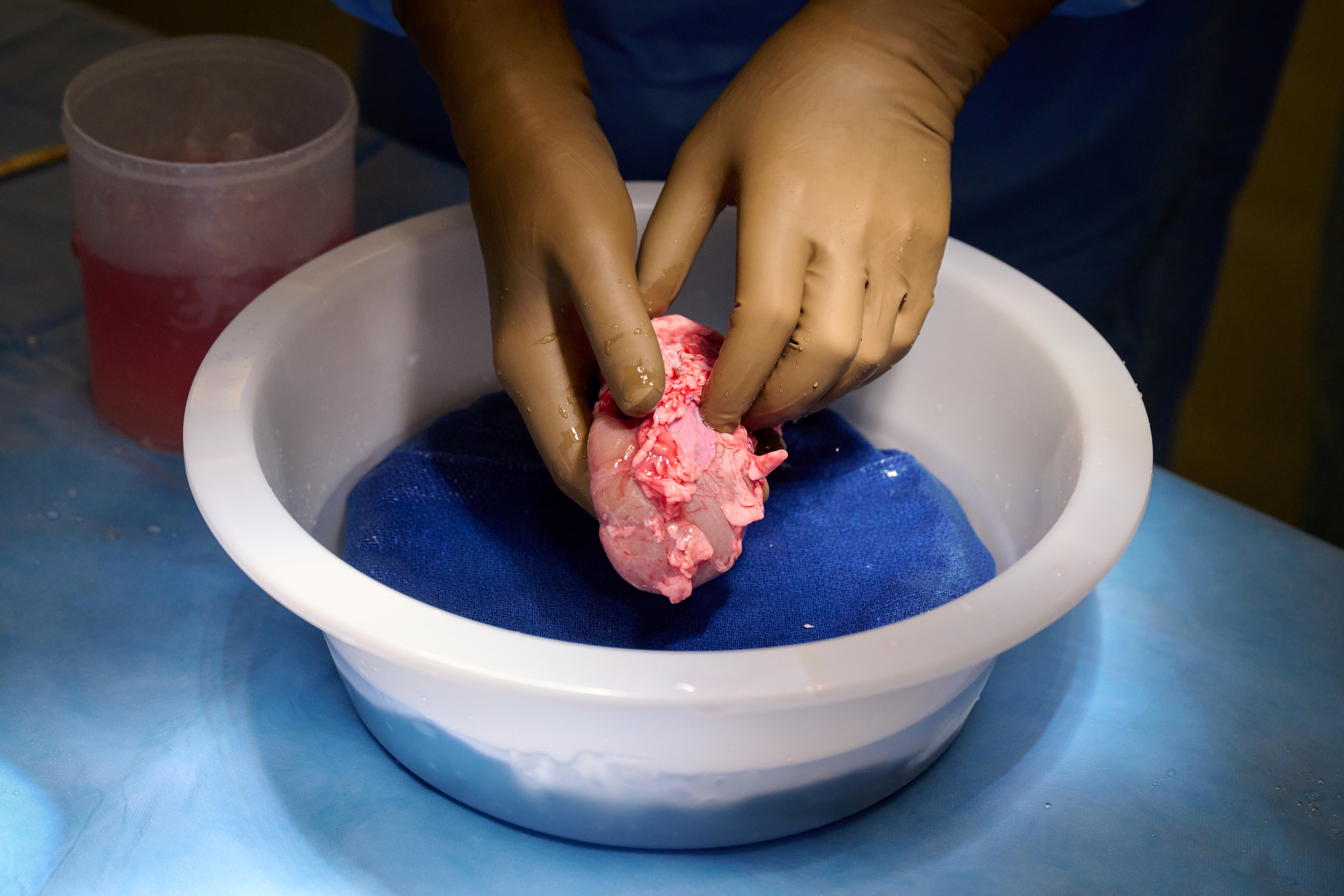
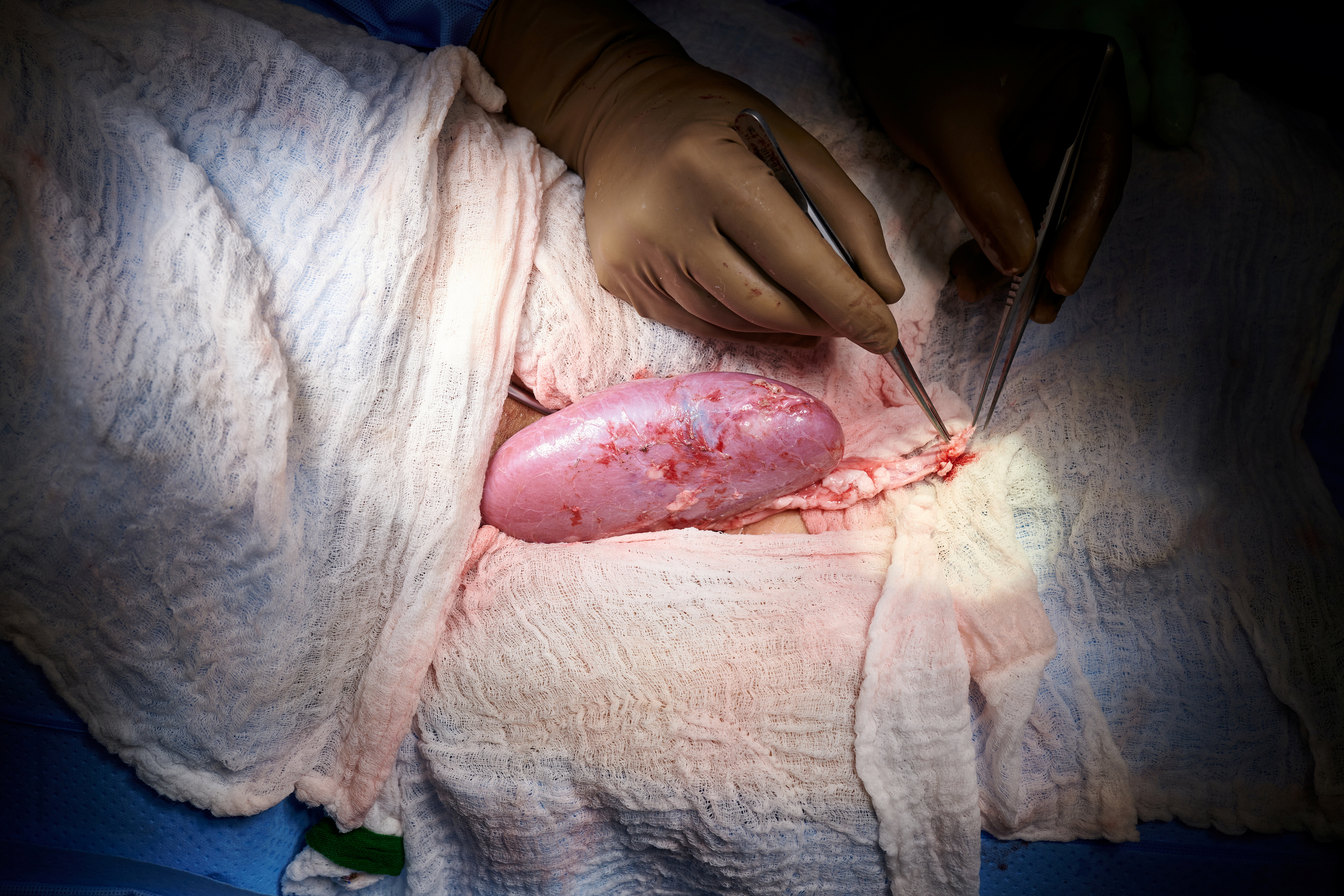
A genetically engineered pig kidney appears healthy during a transplant operation at NYU Langone in New York, U.S., in this undated handout photo. Joe Carrotta for NYU Langone Health/Handout via REUTERS .
This is the first time a kidney from a foreign organism has been transplanted and wouldn’t have been rejected mostly by the host’s immune system, a process known as xenotransplantation.
Xenotransplantation would be considered as any procedure which involves the transplantation, implantation, as well as infusion into a living person as a recipient of either (a), live non-human animal cells, tissues, or organs, or (b) human body fluids, cells, tissues, or organs that have been in contact with live non-human animal cells, tissues, or organs ex vivo. The growth of xenotransplantation is being fueled in part due to the fact that the availability of human donors for clinical transplantation vastly outstrips availability.
Humankind has tried transfusions with animal blood and even a baboon heart transplant, but the patient did not survive. Although pig heart valves, as well as pig intestine-derived heparin, have been discovered to operate well in human recipients with skin grafts, pigs have indeed been discovered to be more compatible with the human body. The surgery, however, required the use of a genetically modified pig in order to be effective.
Pig cells create a sugar known as alpha-galactose, which is found in all mammals except humans. When a typical pig organ gets transplanted into such a person, it contains alpha-galactose, which the human immune system recognizes as a foreign substance. The human body then attacks and finally rejects the transplant. For a variety of health, safety, and logistical reasons, domesticated animals including cows and pigs are considered possible tissue and organ supplies before nonhuman primates such as monkeys. Pigs are selected because they develop fast, generate large litters, and have organs both in infancy or maturity that are equivalent in composition and shape to human organs. They may also be cultivated in microbiologically controlled conditions to meet strict health requirements.
As a consequence of advancements in human-to-human transplantation, xenotransplantation, which had been a topic of research and experimentation for more than a century, began to attract substantial attention in the scientific community there in the 1960s. As in the United States and South Africa, 31 clinical operations involving the transplantation of solid organs of animal donors were done between 1963 and 1993.
In 1963 and 1964, the first studies with chimp kidney transplantation into humans were carried out. Several of the patients who got chimp kidneys survived for nine months after receiving them.
Baby Fae, the child who got a baboon heart in 1984, and Jeff Getty, an AIDS patient who had a baboon bone marrow transplant in 1995, were two of the most well-known xenotransplant surgeries in the previous two decades. Getty rejected the donated marrow nearly quickly, whereas Baby Fae survived with her xenotransplant for 20 days. Getty was still clear of baboon-transmitted viruses and showed no evidence of baboon bone marrow in his system as of October 1998.
The most practical barrier to xenotransplantation is rejection, wherein the recipient’s body assaults the new organ as if it were an infection. Drug therapy, such as cyclosporine, has traditionally been used in organ transplants to suppress recipients’ immune systems, allowing transplanted organs may function without even being attacked or rejected as aliens. When tissue that is not recognized as a human is given to the body in xenotransplantation, a much more severe response mechanism known as “hyperacute rejection” develops. An individual’s immune system goes out to kill the transplanted organ in a couple of minutes. Breeding transgenic pigs is one approach being investigated to circumvent organ rejection. These genetically modified pigs produce human proteins which make it extremely challenging for the body’s immune system to distinguish the porcine organ from that of another animal. A transgenic pig is created by inserting a tiny quantity of DNA (or genetic material) into a fertilized pig egg, which is subsequently implanted into a sow, resulting in the birth of the pig.
Cross-species infection, or the transmission of infectious illnesses from animals to people, is still a hot topic of research, despite the fact that the dangers have decreased. Two of four forms of the pig endogenous retrovirus (PoERV) were shown to be capable of infecting cultured human cells in a test – tube in 1997. An endogenous retrovirus is some kind of virus that is found in all animals’ DNA and is handed on to children without causing damage over generations. This previous article makes no mention of whether viral transmission may occur as a consequence of a transplant or, if it did, whether it would induce illness.
Xenotransplantation has been examined by a number of local agencies throughout the globe and confirmed to be morally acceptable. Whilst also we genuinely think that xenotransplantation may be the solution for hundreds of patients who will require tissue or solid organ transplants, we believe that the industry and the scientific community should proceed with caution in order to ensure the safe development of such potentially life-saving technology.
Sources :
https://www.fda.gov/vaccines-blood-biologics/xenotransplantation
https://www.frontiersin.org/articles/10.3389/fimmu.2019.03060/full














2 Responses
This is really a very nice article, thanks for sharing this article.
I found some very relevant and useful information that I needed.
I hope u keep writing articles like this in the future.
Hi Vibha,
Thanks for your comment. Don’t forget to register on our website for future updates. You can also download our web app available in our website.