In crystallography and mineralogy, a crystal structure is known as a special arrangement of atoms in a crystal. A crystal structure is made out of a motif, a lattice, and a set of atoms arranged uniquely. Motifs are found upon the points of a lattice, which is a set of points recounting occasionally in three dimensions.
The points can be considered as forming indistinguishable tiny boxes, known as unit cells, that occupy the space of the lattice. The lengths of a unit cell edges and the points between them are known as the lattice parameters. The crystal symmetry properties are exemplified in its space group. A crystal’s structure and symmetry has a part in determining large numbers of its properties, for example, electronic band structure, cleavage, and optical properties.
The Seven Crystal Structures
Triclinic System: Every one of the three axes is slanted towards one another, and they are of a similar length. In light of the three slanted points, the different types of crystals are in the paired faces. Some standard Triclinic Systems are Amazonite, Rhodonite, Kyanite, Labradorite, Turquoise, and Aventurine Feldspar.
Orthorhombic System: This involves three axes and is at right angles to one another. These three axes are of various lengths. Given their Rhombic structure, the orthorhombic system comprises different crystal shapes namely double pyramids, pyramids, pinacoids, and rhombic pyramids. Some regular orthorhombic crystals comprise Zoisite, Iolite, Topaz, Tanzanite, Danburite, e t.c.
Monoclinic System: This involves three axes where two are at right angles to one another, and the third axis is slanted. Every one of the three axes is of various lengths. Given the internal structure, the monoclinic system comprises Basal pinacoids and crystals with slanted end faces. A few examples are Howlite, Hiddenite, Vivianite, Gypsum, Petalite, Diopside, Kunzite, and more
Tetragonal Systems: It comprises three axes. The major axis differs in length; it can either be long or short. The two-axis lie in a similar plane and are of a similar length. Given the rectangular inner structure, the shapes of crystal in tetragonal contain trapezohedrons, double and eight-sided pyramids, pyrite, and four-sided crystal.
Cubic System: Each of the three points meets at right angles and is of equivalent length. The crystal shapes of a cubic system dependent on inner construction (square) include Hexaciscoherdron, shape, and octahedron. Examples are Gold, Silver, Garnet, and Diamond.
Trigonal System: Axes and angles in a trigonal system are like Hexagonal Systems. There will be six sides at the base of a hexagonal system (ross-segment of a crystal) and there will be three sides in the trigonal system (base cross-section). Crystal shapes in a trigonal system include Scalenohedral, three-sided pyramids, and Rhombohedra. Some common examples include Calcite, Agate, Jasper, Ruby, Quartz, Tiger’s Eyes, e.t.c.
Hexagonal System: It involves four axes. Among these four axes, three axes are of a similar length and are on one plane. They intersect each other at a point of 60°. The fourth axis intersects the three other axes at 90°s. Crystal states of hexagonal systems comprise Double-Sided Pyramids, Double Pyramids, and Four-Sided Pyramids. Examples are Cancrinite, Beryl, Sugilite, Apatite, and e.t.c.
Crystal structure of steroid 5a-reductase SRD5A reveals preserved steroid reduction instrument
Steroid hormones are fundamental in immune system regulation, stress response, and reproduction in mammals. Steroids with a 3-oxo-Δ4 structure, for example, progesterone or testosterone, are catalyzed by steroid 5α-reductases (SRD5As) to produce their corresponding 3-oxo-5α steroids, which are fundamental for different pathological and physiological cycles. SRD5A2 is as of now a target of clinically significant medications.
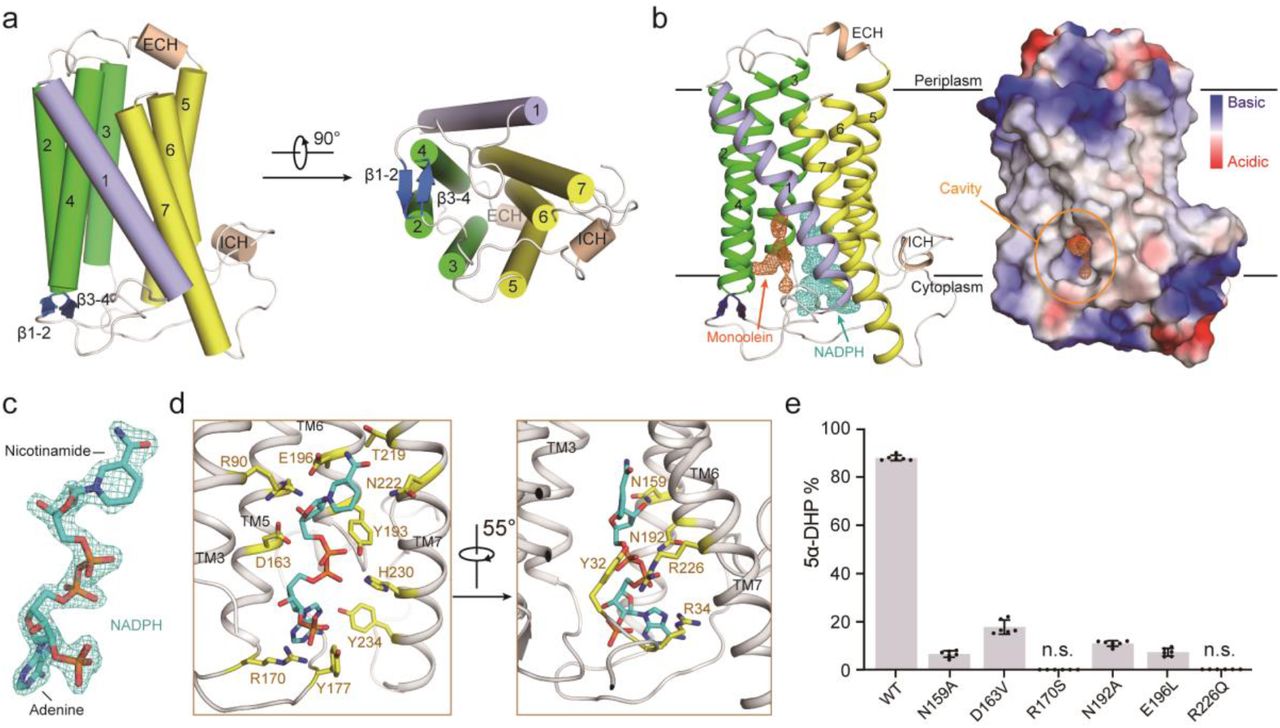
Notwithstanding, the detailed instrument of SRD5A-mediated stays ingenious. PbSRD5A occurs as a monomer containing seven transmembrane segments (TMS). The TM1-4 encircles a hydrophobic substrate-binding compartment, though TM5-7 facilitates cofactor NADPH through a substantial hydrogen bonds network.
Homology-based structural models of HsSRD5A1 and – 2, along with biochemical characterization, define the properties of illness-related mutants, illustrate the substrate-binding pocket of SRD5As, and provide a significant system for further comprehension of the instrument of NADPH mediated steroids 3-oxo-Δ4 reduction. In light of these analyses, the structure of therapeutic atoms focusing on SRD5As with improved specificity and therapeutic viability will not be impossible.
Conductive Nature In Crystal Structures Disclosed At Elaboration Of 10 Million Times
Imperfections, or defects, are basic in crystals. The line defects are a row of atoms that deviate from the typical order. Since dislocations have a similar theme of components as the host crystal, the progressions in electronic band structure at the dislocation core, are regularly just marginally different from that of the host. This is because of symmetry-reduction and strain. The researchers had to look outside the dislocations to locate the metallic line deficiency, where the resulting atomic structure and deficiency composition are greatly different.
An alumni understudy in the Department of Chemical Engineering and Materials Science, Hwanhui Yun stated that they easily detected these line defects in the high-resolution scanning transmission electron microscopy pictures of these BaSnO3 thin films as a result of their special atomic setup.
For this research, BaSnO3 films were developed by molecular beam epitaxy (MBE) in a lab at the University of Minnesota Twin Cities. Molecular beam epitaxy (MBE) is a strategy to create high-quality crystals. Metallic line defects noticed in these BaSnO3 films engender along the film growth direction, which implies specialists can possibly control how or where line defects appear and conceivably engineer them as needed in savvy windows, touchscreens, and other future technologies that request a combination of conductivity and transparency.
Crystal structures in super slow motion
Laser beams can be utilized to change the properties of materials in an incredibly exact manner. This principle is as of now generally utilized in technologies, for example, rewritable DVDs. Notwithstanding, the fundamental processes typically occur at such unfathomably fast paces and at a particularly limited scale that they have so far escaped direct observation. Specialists at the Max Planck Institute (MPI) for Biophysical Chemistry and the University of Göttingen and have now figured out how to film, unexpectedly, the laser transformation of a crystal structure with nanometre resolution and in slow motion in an electron microscope.
Thomas Danz and Professor Claus Ropers took advantage of a unique property of a material comprised of atomically thin layers of tantalum and sulfur atoms. At room temperature, its crystal structure is contorted into a little wavelike structure and a “charge-thickness wave” is created. At higher temperatures, a stage change happens in which the original minuscule waves out of nowhere disappear. The electrical conductivity likewise changes radically, a fascinating effect for nano-electronics. In their research, they generated this stage transition with short laser pulses and recorded a video of the response of charge-density-wave.
Reference:
- https://en.m.wikipedia.org/wiki/Crystal_structure
- https://phys.org/tags/crystal+structure/
- https://www.nature.com/articles/s41467-020-20675-2
- https://www.sciencedaily.com/releases/2021/01/210115155323.htm
- https://www.sciencedaily.com/releases/2021/01/210122154414.htm















