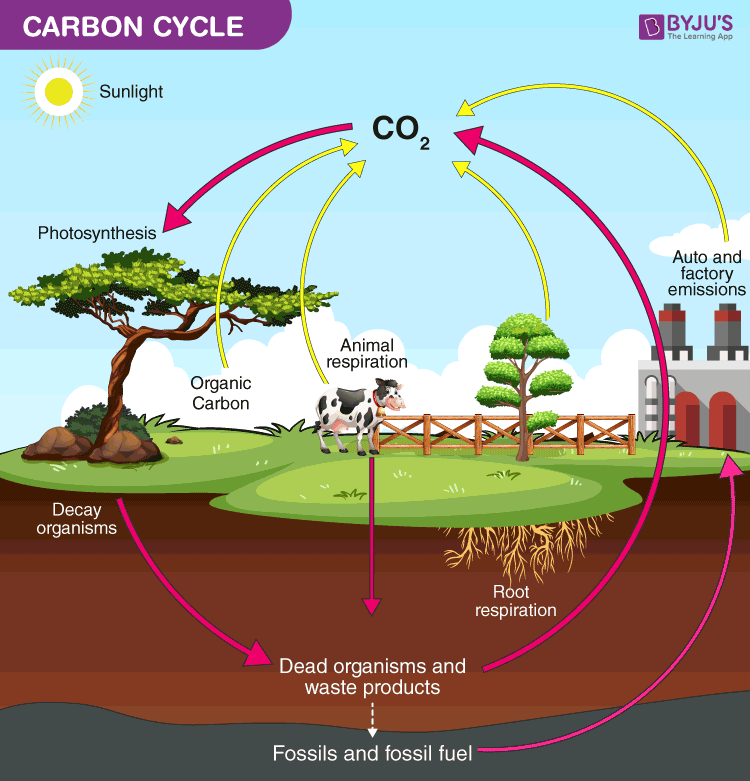
The carbon cycle is referred to as the biogeochemical cycle by which carbon is exchanged among the geosphere, hydrosphere, biosphere, pedosphere, and atmosphere of the Earth. Carbon is the primary segment of biological compounds just as a significant segment of numerous minerals, for example, limestone. Alongside the water cycle and the nitrogen cycle, the carbon cycle includes an arrangement of events that are crucial to making Earth fit for sustaining life. It depicts the movement of carbon as it is reused a lot all through the biosphere, just as long haul cycles of carbon sequestration to and discharge from carbon sinks.
Carbon cycle, in science, course of carbon in different structures through nature. Carbon is a constituent of every natural compound, many of which are basic to life on Earth. The origin of the carbon discovered in living matter is carbon dioxide, which is in the air or decomposed in water. Terrestrial green plants and Algae, which are known as producers are the main agents of carbon dioxide fixation through the cycle of photosynthesis, through which carbon dioxide and water are changed over into basic sugars.
These mixes are utilized by the producers to carry on digestion, the excess being conserved as polysaccharides and fats. The conserved products are then eaten by organisms. This is from protozoans to human beings, which convert them into different structures. Afterward, animals directly add CO2 to the atmosphere as a result of respiration.
Importance of Carbon Cycle
The carbon cycle is tied to other elements and compounds available. For example, the carbon cycle is tied to oxygen available in the atmosphere. While releasing oxygen, Plants take carbon dioxide from the air and used it to make glucose. This happens during photosynthesis
The carbon cycle defines how the segment carbon moves between the Earth’s hydrosphere, air, biosphere, and geosphere. It is important for the below explained reasons:
- Perceiving how carbon moves make us fathom natural cycles and factors that influence them.
- One form of carbon takes is the gas carbon dioxide, CO2. High levels of carbon dioxide secure the Earth, making temperatures rise. Understanding how carbon dioxide is acclimatized and conveyed assist us in understanding the environment and predict an unnatural climate change.
- Carbon isn’t in balance, so it’s fundamental to acknowledge where it is being taken care of and conveyed. The percentage at which carbon is stored in living organisms isn’t the same as the rate it is returned to the Earth. There is approximately a hundred times more carbon in the living matter than in the Earth. Burning fossil fuels releases immense proportions of carbon into the atmosphere and to the Earth.
- The carbon cycle is joined to the availability of various compounds and elements. For example, the carbon cycle is connected to the availability of oxygen in the atmosphere. During photosynthesis, carbon dioxide is taken from the air and utilized to create glucose, while releasing oxygen.
The Changing Carbon Cycle
Today, the carbon cycle is evolving. People are moving more carbon into the atmosphere from different pieces of the Earth framework. More carbon is moving to the air when petroleum products, similar to coal and oil, are burned. More carbon is moving to the air as people get rid of forests by burning the trees. Burning wood discharges carbon into the atmosphere that had been put away in the tree.
The majority of the carbon in the atmosphere is in atoms of carbon dioxide (CO2). Carbon dioxide is a greenhouse gas that allows heat to be held in the atmosphere. By expanding the measure of this greenhouse gas in the air, Earth is getting warmer.
Carbon dioxide spends quite a while, up to many hundreds of years, in the atmosphere, so regardless of whether individuals quit adding carbon dioxide to the air currently Earth would keep on warming. The carbon can gradually move once again into the biosphere, taken up by plants as they photosynthesize. It can likewise move into the seas. What’s more, it very well may be put away in rocks of the geosphere like limestone. Researchers are presently examining these cycles and others that move carbon out of the environment.
The carbon cycle has changed all through the years of Earth’s history. In any case, prehistoric changes occurred for various reasons. The measure of carbon dioxide in Earth’s climate expanded before and again now, during the Devonian period of the Paleozoic for instance, given volcanic emissions. Volcanoes discharge more than ash and lava. They additionally send gases, similar to carbon dioxide into the atmosphere. Today the measure of volcanic ejections is little compared with other times in the past, yet the centralization of carbon dioxide in the atmosphere is very high since individuals are burning forests and fossil fuels.
Effect of Caborn Cycle
Atmosphere
It is huge that so much carbon dioxide remains in the air since CO2 is the main gas for controlling Earth’s temperature. Carbon dioxide, halocarbons, and methane are greenhouse gases that ingest a wide scope of energy—including infrared energy (heat) erupted by the Earth—and afterward re-produce it. The re-produced energy goes out in every direction, yet a few re-visitations of Earth, where it warms the surface. Without greenhouse gases, Earth would be frozen – 18 degrees Celsius. With such a large number of greenhouse gases, Earth would resemble Venus, where the greenhouse atmosphere keeps temperatures around 750 Fahrenheit.
Since researchers know which frequencies of energy every greenhouse gas absorbs, and the gas concentration in the atmosphere, they can figure how much each gas adds to warming the planet. Carbon dioxide causes around 20% of Earth’s greenhouse impact; water vapor represents around 50%, and clouds represent 25%. The rest is brought about by little particles and minor greenhouse gases like methane.
Ocean
Ocean fermentation influences marine organisms in two places. In the first place, carbonic acid responds with carbonate ions in the water to create bicarbonate. In any case, those similar carbonates are what shell-building creatures like coral need to make calcium carbonate shells. With less carbonate available, the creatures need to use more energy to assemble their shells. Therefore, the shells wind up being more slender and more delicate.
In the second part, the more acidic water is, the better it breaks up calcium carbonate. Over the long haul, this reaction will permit the ocean to absorb overabundance carbon dioxide because more acidic water will disintegrate more rock, discharge more carbonate ions, and increase the ocean’s ability to assimilate carbon dioxide. Meanwhile, however, more acidic water will disintegrate the carbonate shells of marine organisms, making them hollowed and weak.
Land
With more atmospheric carbon dioxide that can convert to plant matter in photosynthesis, plants had the option to develop more. This expanded development is alluded to as carbon fertilization. Models anticipate that plants may develop somewhere in the range of 12 to 76 percent more if atmospheric carbon dioxide is multiplied, as long as nothing else, similar to water deficiencies, restricts their growth. Notwithstanding, researchers don’t have the foggiest idea of how much carbon dioxide is increasing plant growth in reality since plants need more than carbon dioxide to develop.
A portion of the changes in carbon absorption is the consequence of land-use decisions. Farming has gotten considerably more escalated, so we can develop more food on less land. In high and mid-latitudes, relinquished farmland is returning to the forest, and these forests store substantially more carbon, both in soil and wood, than crops would. In numerous places, plant carbon is prevented from entering the atmosphere by extinguishing wildfires. This enables woody material -that stores carbon- to develop.
For detailed Knowledge on this topic, visit: https://earthobservatory.nasa.gov/features/CarbonCycle
Reference:
- https://en.m.wikipedia.org/wiki/Carbon_cycle
- https://www.britannica.com/science/carbon-cycle
- https://scied.ucar.edu/learning-zone/climate-change-impacts/changing-carbon-cycle
- https://science.jrank.org/pages/1207/Carbon-Cycle-Importance-carbon-cycle.html














