Credit: John Innes Centre
A new finding reveals how the number and position of genetic exchanges in sex cells, such as pollen and eggs in plants and sperm and eggs in humans, are determined.
The development of crossovers, which allows DNA to be exchanged between pairs of homologous chromosomes(COs), is a key component of meiosis. COs are critical to promoting consistent division of chromosomes during meiosis I and the implementation of genetic variation. The DSBs will be produced when chromosomes are structured along the meiotic axis into threadlike arrays of chromatin loops through a reparation of the programmed DNA double-strand breaks (DSBs).
In meiosis, chromosomes swap huge portions of DNA when sex cells occur. This ensures that every new cell has its distinct genetic structure, explaining why no two siblings are ever fully genetically similar apart from identical twins. These DNA or cross-sectional exchanges are crucial to generate genetic variety, drive evolution, and strictly manage their frequency or position in chromosomes.
In most species, the DSBs exceed COs, most of which are repaired through the creation of recombination intermediate (RI) joint molecules channeled through a reparation mechanism that is not cross-over. The relative locations, and hence numbers, of RIs that proceed to form COs, are closely restricted: CO creation at one chromosomal location hinders the development of further COs in the immediate vicinity. The crossover interference (COI) phenomenon has been discussed for the first time over a century ago and is common in eukaryotes.
Dr. Chris Morgan highlights the importance of this phenomenon as the co-first author of a study: Positioning cross-over has profound consequences for development, fertility, and selective reproduction. By learning about the mechanics for crossover positioning, they will likely be able to find ways to change crossover positioning to improve existing breeding technology for plants and animals.
Despite over a century of inquiry, the cellular mechanism that controls where, and how many, crossovers arise has remained essentially unexplained, a mystery that has interested and frustrated many famous scientists. The expression “crossover interference” was coined in 1915 and captures the finding that when a crossover happens at one point on a chromosome, it hinders the creation of crossovers nearby.
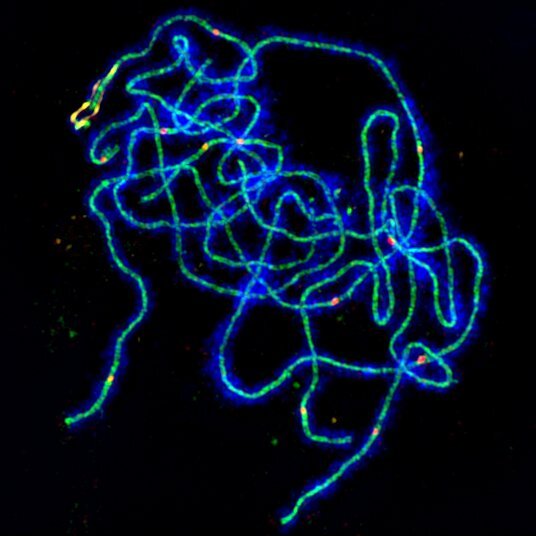
A team of John Innes Centre researchers has solved a century-old mystery by recognizing a mechanism that ensures crossover numbers and positions are ‘just appropriate’: not too few, not too many, and not too close together, using a cutting-edge combination of mathematical modeling and ‘3D-SIM’ super-resolution microscopy.
The scientists analyzed the action of a HEI10 protein that is crucial to meiosis crossover. The cellular abundance of the protein known to modify CO quantities in Arabidopsis is changed, the protein having a conserved E3-ligase domain. Genetically, HEI10 over-expression was demonstrated to weaken COI. However, the impact of HEI10 dose on the positioning of CO along prophase I chromosomes has yet to be investigated cytologically. HEI10 appears as distinct foci along fully-synapsed pachytene chromosomes during meiotic prophase I, apparently in numbers that correspond to MSH4-labeled RIs. From early to late-pachytene, the number of these foci decreases dynamically (from >100 to 15 foci per cell), with late-pachytene foci colocalizing with CO-designated locations identified by MLH1. It’s intriguing to think that the continuous decrease in HEI10 foci that happens at the visible RI to CO transition controls CO positioning directly.
To put this theory to the test, the behavior of HEI10 we examined using super-resolution microscopy. As a result, it was discovered that HEI10’s behavior is compatible with coarsening, and it was argued that HEI10’s coarsening dynamics are the principal driver of CO positioning and COI in Arabidopsis. Next, a modeling technique was used to deduce the molecular basis of HEI10 coarsening by comparing computational simulations of HEI10 coarsening dynamics with 3D-SIM measurements of HEI10 intensity in fixed pachytene cells. In both wild-type and plants with varying HEI10 doses, it was discovered that the simulations are capable of recapitulating and predicting a variety of experimental results. As a result, we argue that a diffusion-mediated coarsening process can explain HEI10-controlled CO positioning and COI.
This measurement was then compared to a mathematical model simulating this grouping based on the distribution of HEI10 molecules and simple principles. Many experimental findings were explained and predicted by the mathematical model, including the fact that crossover frequency could be reliably changed by merely changing the amount of HEI10.
The co-First author Dr. John FoZard explained that they have shown that the data from the super-resolution images of the reproductive cells of Arabidopsis is compatible with a mathematical ‘crossover patterning diffusion-mediated coarsening’ model of Arabidopsis.
The research leverages the legacy of plants as model organisms in the John Innes Center to investigate preserved and fundamental aspects of genetics. J.B.S Haldane and Cyril Darlington examined this identical phenomenon in the 1930s. The model also supports forecasts made in the 1970s by Robin Holliday, another prominent JIC alumnus.
Professor Martin Howard, a corresponding author, adds that this work is a prime illustration of interdisciplinary research, which required advanced experimentation and mathematical modeling to uncover the core of the mechanism. He mentioned that one fascinating way forward would be to evaluate if our model can explain crossover patterning successfully in other creatures.
This research is particularly important for cereal crops like wheat, in which cross-over is mostly limited to certain chromosome regions, thus hindering plant breeders from having full genetic potential for these plants.
References:
Research paper: Diffusion-mediated HEI10 coarsening can explain meiotic crossover positioning in Arabidopsis, Nature Communications (2021). DOI: 10.1038/s41467-021-24827-w
- https://www.news-medical.net/amp/news/20210803/Researchers-solve-century-old-mystery-by-identifying-how-sex-cells-get-the-right-genetic-mix.aspx
- https://phys.org/news/2021-08-sex-cells-genetic-interdisciplinary-approach.html?utm_source=webpush&utm_medium=push














